Instrumentation Capabilities at Biola
Biola’s School of Science, Technology and Health provides state-of-the-art instrumentation to support high quality teaching and research environments. This equipment is used in specific chemistry classes, but is also available for use by all SSTH students, including biochemistry, biology, chemistry, allied health students and more.
All equipment can be viewed within the chemistry labs on the third floor of the LIM Center. In addition to access through the lab classes mentioned above, students can also get training to use the equipment through directed research with our research-active faculty in CPE (CHEM 480). As these instruments are specialty instruments, students will have to receive training through a faculty mentor prior to use. For more information, please contact the CPE department.
Chemistry Labs
The B.S. in Chemistry program in the School of Science, Technology and Health at Biola University is well-equipped to support high quality teaching and research. Note that while these instruments are used in specific chemistry classes, they are accessible by all School of Science, Technology and Health students. See instrument description below.
NMR Spectroscopy: (1) Magritek Spinsolve 80 MHz NMR (2) PicoSpin NMR

NMR Spectroscopy is an analytical tool used to determine the structure of a molecule. Briefly, a sample is dissolved in a solvent and placed in an external applied magnetic field, where it is exposed to radio waves. Depending on the environment of the atom (specifically, nucleus), different amounts of energy will be absorbed at different wavelengths. This information is plotted as intensity vs. chemical shift values. Analysis of the NMR spectrum will help us to determine the structure of the molecule. Note: A nucleus’s environment consists of the different atoms and groups of atoms surrounding it; thus, different nuclei will have different chemical shift values depending on their environment. This is the key behind structural determination. Students will use this instrument in Organic Chemistry Lab, CHEM 311 and CHEM 312, as well as directed research.
UV-Vis Spectroscopy: Agilent Cary 3500 Compact Peltier UV-Vis System

UV-Vis Spectroscopy is an analytical technique used for both qualitative and quantitative analysis, and is one of the most common absorption spectroscopy tools available. It is routinely used to analyze various analytes, including biological macromolecules, transition metal ions, aromatic compounds, or highly conjugated organic molecules. Our brand-new UV-Vis spectrophotometer boasts a dual beam for simultaneous analysis of the blank and the sample, and a 250 MHz xenon flash lamp, which gives the instrument a wavelength range of 190 – 1100 nm and scans at 150,000 nm/min. Students will use this instrument in directed research courses as well as Analytical Chemistry Lab, CHEM 350, and Physical Chemistry Lab, CHEM 406.
FTIR Spectroscopy: MIDAC FTIR Spectrophotometer (M4000-E and PRS)
Fourier-Transform Infrared Spectroscopy is another industry workhorse in the chemical sciences. Like its UV-Visible counterpart, it is used for both qualitative and quantitative analysis, and is another common absorption spectroscopy tool. The wavelength range of our two instruments is from 600 cm-1 - 4000 cm-1, which excites vibrational motion in a variety of functional groups within our molecules of interest. IR spectroscopy features prominently in organic chemistry, because it is used to provide information about the functional groups that are present in our samples for chemical identification. Students will use this instrument in Organic Chemistry Lab, CHEM 311 and CHEM 312.

Inductively-Coupled Plasma Mass Spectrometry: Agilent 8900 ICP-MS Triple Quad
Inductively coupled plasma mass spectrometry (ICP-MS) is a mass spectrometry technique for the purpose of elemental analysis. It features an inductively coupled plasma, which ionizes the sample. ICP-MS is commonly used for the identification and quantification of metals, and sometimes non-metals, in liquid samples, even at very low concentrations. The mass spectrometer gives the instrument the ability to distinguish between chemical isotopes. The ICP-MS is commonly used in water quality, food science, and the semiconductor industry. The instrument is part of Analytical Chemistry, CHEM 350, but can also be used in directed research projects.



High-Performance Liquid-Chromatography Mass Spectrometry: Waters Alliance e2695, with PDA and QDa
High-Performance Liquid-Chromatography Mass Spectrometry (HPLC-MS) is an analytical tool that can be used to separate and identify chemical compounds within chemical mixtures. Typically, a liquid sample mixture is separated by passing the mixture through a chromatography column using a mixed aqueous-organic solvent. Compounds are separated based on their affinity for the stationary phase (on the chromatography column), with higher-affinity molecules showing up at later times on the chromatogram. The HPLC is equipped with two detectors: a photo-diode array detector (PDA, for UV spectroscopy), and quadrupole mass spectrometer (QDa). The HPLC-MS is found commonly in all chemical industries, including food sciences and pharmaceutical sciences. Students will use this technique in Organic Chemistry Lab (CHEM 311 and CHEM 312), as well as Analytical Chemistry (CHEM 350) and directed research.



Gas Chromatography – SRI 310C and 8610C Chromatographs
Gas chromatography (GC) is an analytical tool that can be used to separate and identify chemical compounds within chemical mixtures. Typically, a liquid sample mixture (of volatile compounds) is separated by passing the mixture through a chromatography column using a carrier gas (typically N2 or He). The GC is equipped with a flame ionization detector (FID). The GC is found in all chemical industries, but has historically been very prominent in the oil and gas industry. Students will use this technique in Organic Chemistry Lab (CHEM 311 and CHEM 312), as well as Analytical Chemistry (CHEM 350) and directed research.



Nursing Skills Labs
The B.S.in Nursing program in the School of Science, Technology and Health at Biola University is well-equipped to support high quality teaching and safe, realistic clinical experiences.
Manikins
Nursing Anne Simulator, SimMan, and SimMom are high-fidelity manikins designed for scenario-based training in the care and management of a wide range of in-hospital patients. These versatile simulators support clinical training across multiple specialties, including women's health, obstetrics, postpartum care, wound assessment and care, general patient assessment, and both emergent and urgent care situations.
These manikins feature realistic anatomy and responsive functions. They can speak, seize, and accommodate a variety of clinical procedures such as CPR, medication administration, nasogastric tube insertion, tracheostomy care, suctioning, and both intramuscular and intravenous injections. SimMom is specifically designed to simulate childbirth, allowing students to experience labor and delivery scenarios.
Learners can also auscultate normal and abnormal heart sounds, lung and bowel sounds, and fetal heart tones, providing realistic diagnostic practice. Throughout the nursing program, students will engage with these manikins in the Nursing Skills Lab as part of every clinical course.
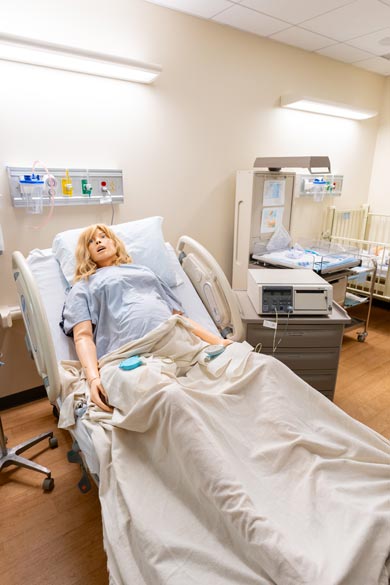
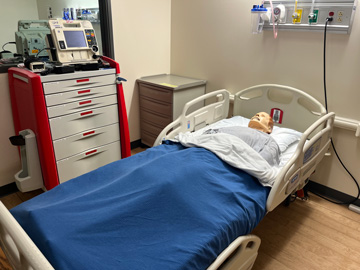
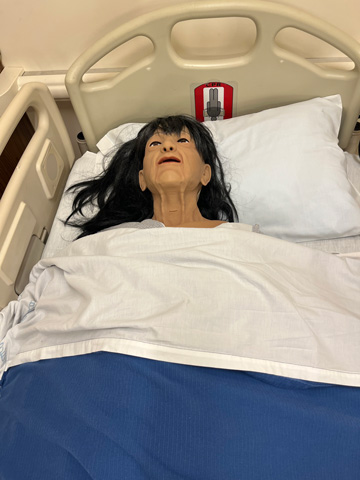
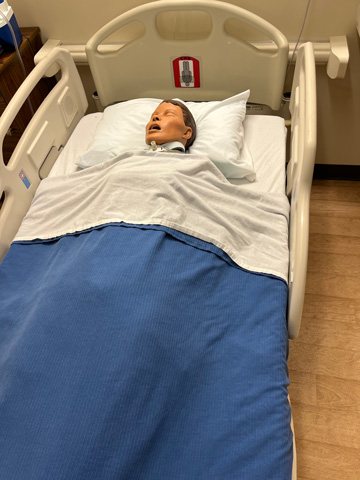
Patient Monitor

A laptop-operated PC is used in conjunction with our high-fidelity manikins to simulate real-time patient monitoring. Students will utilize this system in the Nursing Skills Lab throughout their clinical courses, progressively building their assessment skills and clinical decision-making abilities. The monitors closely resemble those used in hospital settings, enhancing realism and allowing students to interpret dynamic changes in patient status as clinical scenarios unfold.
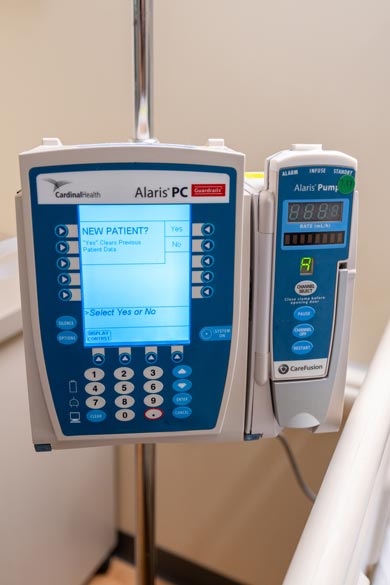
Pumps
Alaris, B. Braun, and Plum IV pumps are widely used in local hospitals, providing students with realistic and accurate preparation for clinical practice. Our Alaris IV pump modules are set up with tubing and fluid bags to simulate the continuous or intermittent delivery of fluids, medications, blood, and blood products to adult, pediatric, and neonatal patients.
In addition, we offer Kangaroo feeding pumps to support training in the administration of enteral nutrition, reflecting the types of care students will encounter during hospital rotations.
Students have the opportunity to practice using these devices during structured clinical skills classes and Open Skills sessions, reinforcing competence and confidence in IV and enteral therapy techniques.
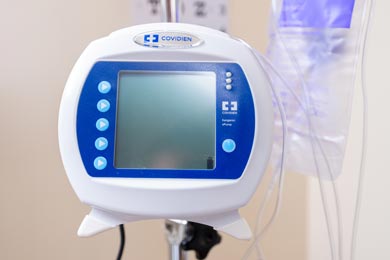
Resuscitation Quality Improvement (RQI) Machine
This American Heart Association digital resuscitation program is the gold standard for delivering high-quality CPR training and continuous resuscitation improvement. RQI will verify students’ competence each quarter through short eLearning and skills activities. The eLearning includes cognitive assessment activities which will ask you to apply cognitive knowledge by treating a patient within the steps of an algorithm. The skills simulation station will provide students’ with real-time audio and visual feedback as they proceed through ventilations and compressions on adult and infant manikins. A debrief will score students’ on hand placement, rate, recoil, chest compression fraction, and depth of compressions, as well as ventilation feedback so you will know what you did well and what you may need to improve.
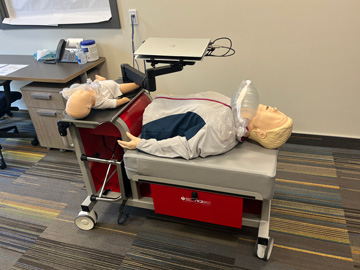
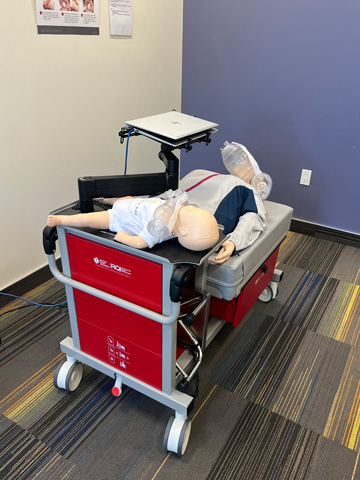
IV Arms & Wearable IV Arms
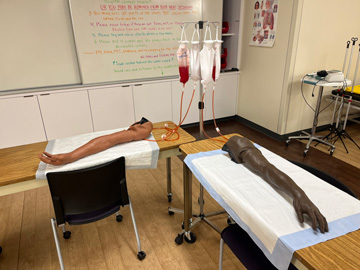
There are multiple high fidelity IV arms that have blood flow in order to provide accurate feedback and more realistic implementation of peripheral IV insertion and blood draws.
Chester Chest

Chester Chest provides students with hands-on opportunities to practice a variety of central line care procedures, including PICC dressing changes, central line cap changes, port access, and central line blood draws.
Bariatric Manikin
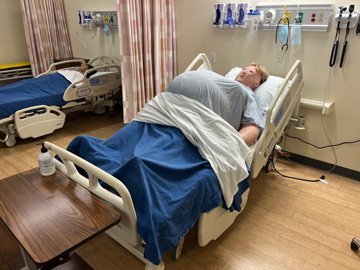
With this manikin, students can practice a wide range of essential nursing skills, including range of motion exercises, perineal care, and patient transfer and lifting. The manikin also allows for training in pressure injury identification, skin assessments, blood pressure reading, and other basic patient care procedures.
Communications Science Disorders
Nursing Anne Simulator
The Nursing Anne Simulator is a lifelike simulator that is used to demonstrate what Speech-Language Pathiologists (SLPs) would do with a patient in the medical setting, such as conducting an instrumental FEES exam (endoscopy). When powered on, the simulator breathes, blinks, imitates human-like movements, and also has pre-recorded live vocal sounds to better simulate a realistic patient. Professors first demonstrate how to conduct the specific simulation on the simulator and students will be given an opportunity to practice under supervision. Students will use this instrument for labs in CODS 432 Dysphagia and MSLP 537 Advanced Dysphagia.
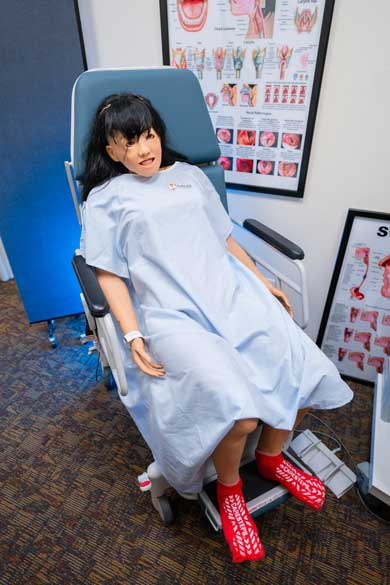
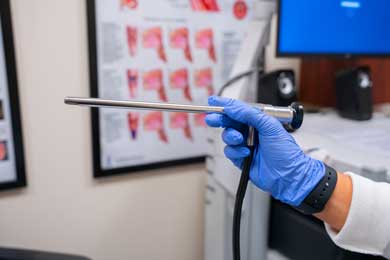
Speech Lab: Laryngeal Strobe (9400)/Voice Swallow Station/Endoscope
Using the Nursing Anne Simulator, students will learn and practice how to use a flexible endoscope to assess swallowing and voice abilities. An endoscopy is a procedure done to visually examine what happens in your mouth and throat during swallowing and vocalizing. The endoscope is a long, flexible tube that has a tiny camera at the end to visualize the area for an etiology–it connects to a computer and video monitor for visualization. This exam is often recorded so that the SLP can watch it again later. Having an endoscopy done allows the SLPs to visualize if food goes into your airway instead of the patient’s stomach, what parts of the patient’s mouth and throat are not functioning properly, what kinds of foods are safest for the patient to swallow, and what positions or strategies can help the patient swallow better. It also allows the clinician to see how the vocal cords are functioning during speaking. Students will use this instrument for labs in CODS 432 Dysphagia, CODS 444 Voice Disorders, MSLP 537 Advanced Dysphagia, and MSLP 542 Advanced Voice Disorders
-Voice-Swallow-Station.jpg)
Voice SonaSpeech
Voice SonaSpeech is a software that is used in therapy to measure the sound waves for voice, articulation, fluency and motor speech patients. This software presents real-time visual feedback displays, games and graphic rewards to motivate children in therapy, seven separate modules for assessment and treatment for communication disorders patients, and quantitative measurements to track client progress. After being given a detailed instruction on how to work this software, students may be asked to demonstrate its use on themselves or with a peer under supervision. Students will use this instrument in CODS 444 Voice Disorders and MSLP 542 Advanced Voice Disorders.
Audiometers (MAICO MA25, MAICO MA25, MAICO MA25 II, INTERACOUSTICS AS608, INTERACOUSTICS AS608, INTERACOUSTICS AS608, G S I 17, G S I 18, G S I 17, INTERACOUSTICS AS208
Audiometers are instruments that are used to test one’s ability to hear sounds. Using an audiometer, professors and students can perform a pure tone testing. For this test, earphones are attached to the audiometer and if properly trained, pure tones of a specific frequency and volume are delivered to one ear at a time. When the testing is done, it will either yield a pass or fail result. Under supervision and with guidance from our professors, students will be using an audiometer to test the hearing of peers and others to build up their clinical repertoire. Students will use this instrument for labs in CODS 425 Introduction to Audiology, CODS 424 Aural Rehabilitation, CODS 429 Clinical Practicum, CODS 441 Diagnostic Methods, MSLP 601 Diagnostic Team, and MSLP 547 Audiology and Aural Rehabilitation for SLPs.

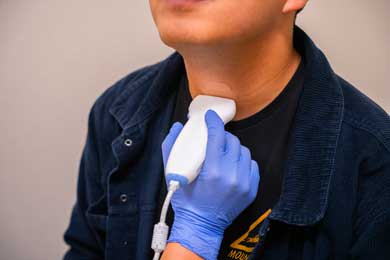
Ultrasound (SP-L01)
The ultrasound instrument is used in therapy for clients with speech sound errors. Ultrasound biofeedback can provide these clients with additional knowledge about their tongue movement and placement when producing different sounds. This instrument has its own software that is accessed when connected to a computer. Real-time tongue movements can be viewed when the transducer is held underneath our chin. Students can use this instrument for labs in CODS 439 Speech Sound Disorders and MSLP 536 Advanced Speech Sound Disorders.
Clinical Audiometer
In our soundproof audiological suite is a clinical audiometer. Similar to our portable audiometers in the speech lab, professors and students can test a patient’s hearing. In addition to pure tone testing, this audiometer can help differentiate if hearing loss is happening in the outer ear or inner ear. Under supervision and with guidance from our professors, students will be using an audiometer to test the hearing of peers and patients. Students will use this instrument for labs and clinical practice in CODS 425 Introduction to Audiology, CODS 424 Aural Rehabilitation, CODS 429 Clinical Practicum, CODS 441 Diagnostic Methods, MSLP 601 Diagnostic Team, and MSLP 547 Audiology & Aural Rehabilitation for SLPs.

Biology Labs
qPCR Quantstudio 6

The QuantStudio 6 real-time PCR system is designed to conduct quantitative PCR (qPCR) to compare gene expression across multiple samples. The gene of interest can be measured after each round of amplification through a fluorescent signal. The systems can detect differences as small as 1.5-fold in singleplex reactions with 10 logs dynamic range. Quantstudio then allows easy visualization of results through an intuitive user-interface and is capable of generating presentation-ready figures. The system is currently used in multiple research labs and student projects through the BIOS 450 Directed Research course.
Azure Biosystems Sapphire Biomolecular Imager

The Azure Sapphire Biomolecular Imager is a highly versatile, next generation laser scanning system. It uses up to four solid state lasers (blue/488 nm, green/520nm, red/658 nm, and near infrared (NIR)/784nm) as excitation sources, which helps detect subtle changes and increase image quality. In addition, it offers three detection modes: fluorescence, chemiluminescence, and phosphor, enabling a wide range of applications. This system, which allows for improved multiplex fluorescent detection, higher sensitivity for lower limits of detection and broad linear dynamic range for accurate quantitation, can be used to scan blots, gels, slides, and microplates. Students may use this equipment in the BIOS 450 Directed Research course.
Agilent Novocyte Flow Cytometer
The NovoCyte Flow Cytometer is a high-performance benchtop flow cytometer that allows for complex cell analysis. Flow cytometry is a technology that provides rapid analysis of single cells in solution for multiple parameters (such as cell size, granularity, and fluorescence) by utilizing lasers as light sources to produce both scattered and fluorescent light signals that are read by detectors. This flow cytometer can detect many parameters with enhanced sensitivity and resolution and analyze samples in single tubes, multi-tube racks, or 24-, 48-, or 96-well plates. Students may use this instrument in the BIOS 450 Directed Research course.




S3e Cell Sorter
The S3e Cell Sorter is a sophisticated automated cell sorter that is equipped with three lasers and four fluorescence detectors plus forward and side-scatter detectors that sorts cells fast while maintaining high sort purity, recovery and yield. In addition it is able to sort two different defined population of cells at the same time. This equipment is used in our molecular biology research.


Olympus FLUOVIEW Confocal Laser Scanning Microscope
The Olympus FLUOVIEW FV3000 confocal laser microscope features high-resolution optical imaging, depth sensitivity, and speed. The word confocal refers to an image collected from the focal plane only and laser scanning means that laser excitation was used to acquire the images. Whereas a conventional microscope allows you to see as far as the light can penetrate, confocal microscopes use a focused laser beam to scan small areas of the sample point-by-point to capture fine details. This powerful microscope allows us to see visual sections of the samples that can be constructed into a 3-D structure without having to physically section them, aiding the study of neurodevelopment in the zebrafish embryos (<1 mm) while they are undergoing development live. We are actively using this microscope in our BIOS 450 Directed Research course and primarily for undergraduate research to access dynamic cellular and molecular processes in live specimen, tissue sections, and in cell culture.




Cell Culturing Biosafety Cabinets
Cell culturing biosafety cabinets (BSCs) are used to create and maintain a clean work environment when working with tissues, cells, and other potentially infectious materials. Using BSCs protects not only the user, but also the materials and the environment. Air from the BSC passes through a high-efficiency particulate air filter before being exhausted to the environment, effectively trapping all known infectious agents (>99.97% of particles 0.3 µm in diameter). Students will use this equipment in directed research.


Organic Garden
The Biola Organic Garden is a quarter-acre property on Biola’s main campus maintained by the Department of Biological Sciences. Ongoing projects include: seasonal vegetable beds, fruit tree and vineyard management, tilapia fish culture, aquaponic food production systems, chicken egg production and various composting systems. The garden is used as a part of several biology courses, and is also open to volunteers from all majors and departments. Biodiversity, sustainability and organic methods are emphasized.


Physics and Engineering
3-D Printers
3-D printers are responsible for bringing computer-aided designs (CAD) to life via additive manufacturing. Essentially, CADs are sliced up into multiple layers on a software and saved as files that informs the 3-D printers about the specifications of the layers, such as thickness, infill density, and whether to have a base or support. Printed objects always are manufactured from the bottom up as the heated nozzle ejects melted material (e.g. PLA) onto a heated stage. As the material is layered, the layers integrate and cool down to take the form of the printed prototype. We have two types of 3-D printers in the Engineering Prototyping Laboratory: Ultimaker (S3 and S5) and Bambu Lab P1S. Our engineering students can seamlessly translate their designs into printed objects, which are parts that are assembled into a larger prototype.




Meade 16” ACF Schmidt-Cassegrain Telescope
Telescopes are an essential tool in astrophysics to gather light from stars, planets, galaxies, and nebulae for both qualitative study and quantitative analysis. The Lim telescope and dome are computer controlled, making observations reasonably routine. Although the light pollution in Los Angeles is significant, basic studies of stellar objects can be carried out using narrow-band light filters. The telescope is equipped with a Shelyak Instruments LISA spectroscope so we can analyze variable stars. Students may use these instruments in Astronomy, PHSC 110, directed research courses, and the Advanced Physics Lab, PHSC 480.




Atomic Force Microscope

The NanoScience instrument is an atomic force microscope that uses interactions between a sharp tip with molecules on a surface to study surface structures at a nano-meter resolution. It has been used to study cell structures and is capable of working under both dry and wet conditions.
Scanning Tunneling Microscope

This NanoScience instrument uses quantum tunneling to study surface structures on conductors. Due to the sensitivity of the tunneling current to the potential barrier width, this microscope is capable of reading nanometer resolutions.
Supercomputer

This computer is Biola’s home-made Linux cluster. It consists of eight server computers connected through high speed internet and controlled by a master node. It is used for computationally-intensive scientific research that cannot be conducted on personal or regular computers. It has been used for Density Functional Theory calculations and is capable for other computational physics/chemistry/biology studies (such as molecular dynamics calculations).
Epilog Laser Fusion M2 Laser Cutter

This machine utilizes a high-powered laser to both cut and engrave designs with precision and accuracy. The design and specifications of the process are defined in a vector graphics program called CorelDRAW. Two-dimensional CADs are prepared on CorelDRAW and as the laser cutter is connected to a PC via USB, the xy-position of the design and the laser power and speed are all defined by the user. Depending on the laser power and speed, the result would be either cutting or engraving, in which the latter is frequently utilized in tech industries like Apple for personalized engravings on iPhones or AirPods cases. The laser cutter can create designs on a variety of materials including wood, acrylic, textiles, paper, plastic, and much more. Our engineering students have the means to cut specific parts of various materials for their projects or embellish their prototype with customized logos and artwork.
JLW Instruments Mark-10 F505-IMT Tension/Compression Test System

This device uses a pair of compression plates to mechanically acquire load and compression distance data for various materials, which can be used to experimentally determine properties such as toughness and Young’s modulus. The bottom plate is fixed onto the bottom of the tester while the top plate is attached to a motor that is able to move vertically at a user-defined speed. The screen is a miniature computer that interfaces the software and hardware, in which the user delineates the parameters of a test, such as speed, maximum load, data export, and much more. Our engineering students can calculate material properties for their projects, which allows them to conclude the feasibility of material selection.
NI ELVIS III

The NI ELVIS III boards are used in our Circuits & Instrumentation Laboratory. ELVIS stands for the Engineering Laboratory Virtual Instrumentation Suite made by National Instruments. It combines the precision and accuracy of seven commonly used lab instruments including a 4-channel oscilloscope, function generator, digital multimeter, variable power supply, logic analyzer, Bode plotter, and IV analyzer. Students can connect a PC to the ELVIS via USB, build circuits on its detachable protoboard, and conduct experiments conveniently and efficiently. Thus, theoretical concepts can transition to physical implementation through a safe, in-depth, and engaging project-based learning experience.
Voltera V-ONE PCB Printer

The PCB printer is a desktop-sized prototyping tool. It can print traces using conductive ink, drill through-holes, dispense solder paste, and with its built-in reflow bed it heats up the board from bottom, melts the solder, and attach surface-mount components. It only takes an hour or two to make a printed circuit board from the PCB design files on your computer, instead of several weeks if you send the design to a fabrication house. Our engineering students can test their ideas quickly and iterate within hours by using the PCB printers in our Circuits & Instrumentation Laboratory.
Carbide 3D CNC Milling Machine and Router
The Nomad 883 CNC milling machine and the Shapeoko CNC Router are both Computer Numeric Controlled machines made by Carbide 3D. The Shapeoko CNC Router provides a larger cutting area and can handle a wide variety of materials including softer woods and plastics. The Nomad 883 is better suited for harder materials like aluminum and steel. These machines as well as the 3D printers are used by engineering students for rapid prototyping of mechanical structures and enclosures.


Kinesiology
Major Instruments: Exercise Equipment
SkiErg Machine
The SkiErg machine originated in the cross-country skiing world. The Erg part of the name stands for ergometer, which is a device that measures the work you put in. The SkiErg is a great machine to use for aerobic fitness, and students will learn to use it to improve strength and endurance. Under the guidance of expert faculty, students will use the machine to work muscles in the arms such as the pectoralis, trapezius and deltoids; lower extremity muscles like the quads, glutes, hamstrings, back extensors and hip flexors; and abdominal muscles.



Major Instruments: Physiology Measurements
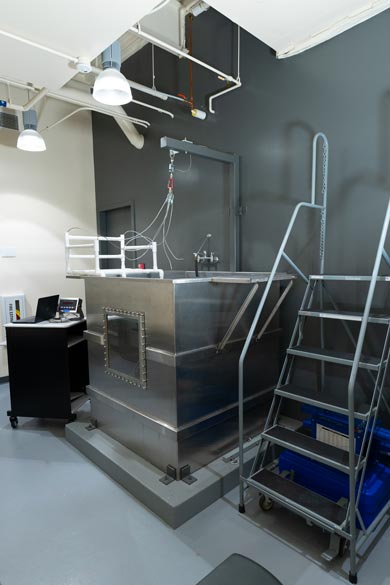
Hydrostatic Weighing Tank
Students in the Kinesiology program at Biola measure hydrodensitometry at Biola University in our state-of-the-art hydrostatic weight measurement tank. Students in our program learn how to measure body composition via gold-standard lab methodologies, with opportunities to practice measuring body density and body fat percentage on each other under supervision.


Cycle Ergometers (Monark 894E and 828E)

Cycle ergometers are a type of stationary bike that students learn to use to measure the amount of energy exerted and exercise completed during pedaling. Cycle ergometers can be used for:
- Performance testing and training
- Power testing and training
- Fitness testing, including Wingate tests, Astrand, YMCA
- Rehabilitation
Technogym treadmills (Excite Med and Runrace)
Designed for conditioning and enhancing sports performance, these treadmills offer the ability to test the limits of athletes and collect data. Students use these to perform exercise tests and learn how to create a personalized exercise program.


Major Instruments: Movement Science Measurements
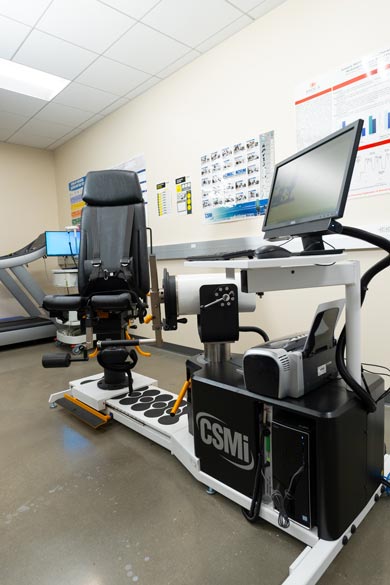
HUMAC-NORM Isokinetic Dynamometer
The HUMAC NORM can measure and improve aspects of human performance in the athletic training room, the clinic and the research lab. Students will use this machine to evaluate muscle strength, power, joint function and proprioception. There are 22 isolated-joint movement patterns, four different modes of resistance (passive, isometric, isotonic and isokinetic) and many different reporting features that students will learn to employ.



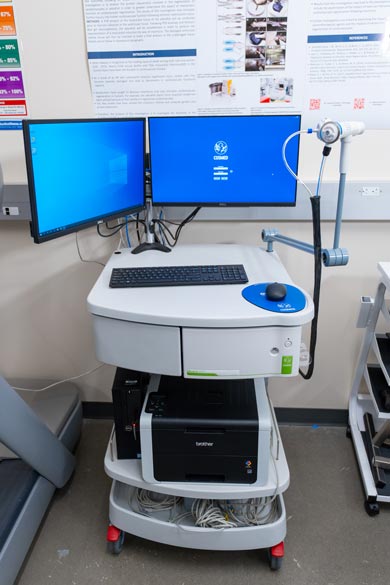
COSMED-Quark: Cardiopulmonary Exercise Testing
Students in the Kinesiology program at Biola learn to perform assessments of a person’s physiological response to exercise. Cardiopulmonary Exercise Testing (CPET) tests the heart (cardiovascular) and breathing (respiratory) systems at the same time during exercise. It measures lung volumes at rest and throughout exercise, as well as blood pressure and the heart’s electrical activity. Biola’s advanced equipment boasts high-quality components and super-fast analyzers to facilitate accuracy, reliability and real breath-by-breath analysis of pulmonary gas exchange, even during high intensity exercise.

AMTI Force Plate & Qualisys Motion Capture System
Force plates are used to characterize biomechanical processes such as walking, running or jumping. The data collected can be used for gait, balance, force, and performance diagnostics in sport or for clinical analyses or in research. The Kinesiology department’s biomechanics lab combines force plate capture with motion capture, to measure the motion of bony segments during various activities. Markers are placed on the subject’s body, which are tracked by special cameras positioned throughout the lab. The software records the joint movement and creates a 3D model of the movement.
